Kelvin’s Thunderstorm
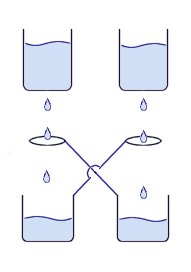 This is a device is a simple method for generating a high voltage charge or static electricity. It works by dripping water through an arrangement of metal containers and loops of wire.
This is a device is a simple method for generating a high voltage charge or static electricity. It works by dripping water through an arrangement of metal containers and loops of wire.
Water, like many molecules is in the form of a dipole. This means that it has a difference in electric charge from one end to the other. The chemical formula for water is H2O, meaning there are two Hydrogen atoms and one Oxygen atom in each molecule.
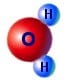 You can see from the image on the right the way that the H2+ is more on one side therefore making an uneven distribution of charge i.e. a dipole. This is what gives water all its interesting properties such as its powerful solvent abilities and its ability to stick to other materials.
You can see from the image on the right the way that the H2+ is more on one side therefore making an uneven distribution of charge i.e. a dipole. This is what gives water all its interesting properties such as its powerful solvent abilities and its ability to stick to other materials.
This experiment works best with pure or deionised water as it is a very good electrical insulator. Tap water contains dissolved ions which become mobile allowing the water to conduct electricity.
As the streams of water droplets pass through the rings, charges are induced making the drips and ring oppositely charged. If a water droplet is made more negative it will help to make the drops on the other side more positive because of the cross connection between the collector cans and the metal loops. This process continues to cause the voltage to rise until the water drops start being deflected by the strong fields or a spark is produced.
The top containers can be made from plastic but it may work better if they are made from metal and connected to ground.






I built one of these in the 70s using 4 large coffee cans, parrafin wax as an insulator under the cans. Heavy copper wire soldered to the can seams and copper pipe soldered to the copper wire. Eye droppers, tubing and tubing valves regulated the flow. Worked great.
Yes both types will work just fine. de-ionized water offers the advantage of having no mobile charge carriers (it is non conductive) therfore energy is not wasted by current flowing in the water.
i do not see why de-ionized water would work better. The operative mechanism is that the water break into droplets under the influence of the electic field in order to trap the charges and accumulate – carry them to their respective buckets. there are more charges in ionized water than de-ionized so i think either will work. If charges can move around better in water the mechanism will probably work better.
Has anyone tried to build this outdoor, using rainwater? You could power UK quite easily…
I believe the reason for wanting the water output to be in droplets, is that a continuous stream would act as a conductor (though, perhaps the deionized water eliminates this concern?)
My instructor showed me one of these at the University of Michigan Electrical Research lab in 1963. He called it the Kelvin Electrostatic Water-Dropping Generator. He used wooden curtain rings wrapped with aluminum tape. The spigots were the lower half of ball-point pen barrels. The flow was adjusted so that the water streams broke into droplets at the ring (I assume for faster charge build-up).
You can create it by distilling (evaporate and collect steam) tap water.
oops! sorry. I just reread the page.
is there a way to deionize water at home?
ok. and one more question: will this work with ordinary tap water, or will it need to be a special, distilled water? Thank you again.
It will work without a leyden jar (a HV Capacitor).
The bottom two containers act like the opposing plates of a capacitor which allows a voltage difference to build up between them. The shape and size of them limits how much energy can be stored so a leyden jar is often used to increase this.
I have googled “Kelvin’s Thunderstorm”, and many of the devices shown have a leyden jar to stor the charge. Is this really necessary? or will it still make a spark anyway? thank you in advance.
I made one of these for science class back in the early 70’s. I got the instructions from a Popular Science Magazine article. It really works though it is hard to see the spark.
Here’s a demonstration of the kelvin’s thunderstorm experiment by prof Walter Lewin. This is an introduction to physics course at MIT.
I’ve never heard of anything more than a table top sized one. I think there are practical barriers to creating a much larger device.
If the charged cans were larger it would mean that they could hold more charge, but they would also have more surface and edges that were discharging energy by corona discharge.
As the charge builds up on the induction rings, the water gets deflected so much that it could pass through the ring and then change direction and hit the side of it. At this point the device would get a little pear shaped 🙂
has any one ever builta kelvin thunderstorm electrostatic generator that that was say one 100 feet high to see how much electricity u can produce with it? or made one that ucould use in a pracatical application?